 between the number of electrons present in bonding and antibonding molecular orbitals. The reason for the unexpected stability of organic compounds in an oxygen atmosphere is that virtually all organic compounds, as well as H 2O, CO 2, and N 2, have only paired electrons, whereas oxygen has two unpaired electrons. Class 11 Chemistry Chemical Bonding and Molecular Structure NCERT. Fortunately for us, however, this reaction is very, very slow. 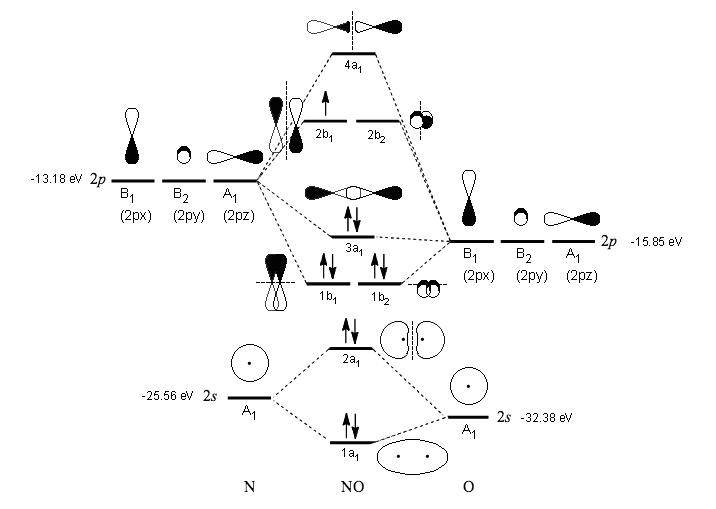
Because Earth’s atmosphere contains 20% oxygen, all organic compounds, including those that compose our body tissues, should react rapidly with air to form H 2O, CO 2, and N 2 in an exothermic reaction. Determine the number of electrons in the s s and p p orbitals. Full video can be found at The magnetic properties of O 2 are not just a laboratory curiosity they are absolutely crucial to the existence of life. Determine the total number of valence electrons. Consequently, it is attracted into a magnetic field, which allows it to remain suspended between the poles of a powerful magnet until it evaporates. 
Readers are strongly encouraged to consult the modiagram package documentation which contains numerous helpful examples to demonstrate its many featuresfar more than we can address in this short article. 
\): Liquid O 2 Suspended between the Poles of a Magnet.Because the O 2 molecule has two unpaired electrons, it is paramagnetic. This article provides a brief introduction to the creation of molecular orbital diagrams in LaTeX using the modiagram package. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
